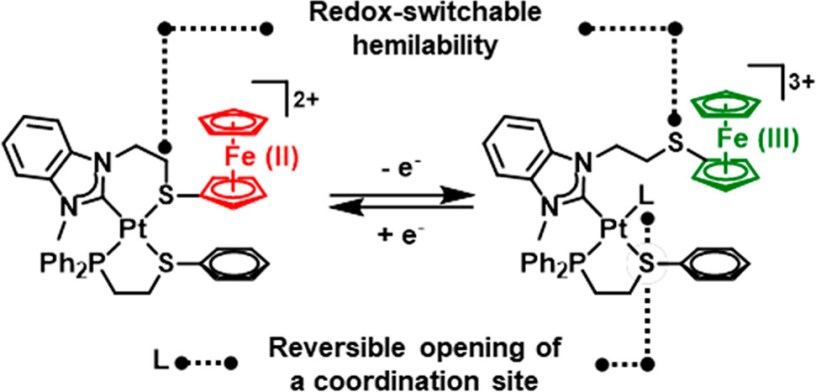
The Weak Link Approach (WLA) is a successful strategy for allosterically controlling the formation of cavities¹ and the access to them² through the action of reversible hemilabile-bond formation around an organometallic center. Thus far, the WLA has been used to mimic biological cavities whose access is controlled chemically as in the scheme shown below which belongs to a previous WLA work published in 2014, my first time involved in the calculation of bond energies for hemilabile groups.
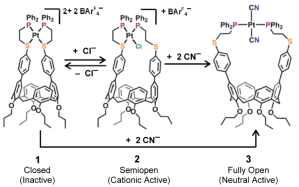
Chiefly developed by the Chad Mirkin group at Northwestern, the WLA has now reached a new milestone in which the allosteric control is further coupled to a redox equilibrium which alters the strength of the hemilabile bonds. These findings are reported in JACS as a communication (DOI: 10.1021/jacs.8b09321). Previous efforts were unsuccessful due to the instability of the oxidized species, which makes regulation challenging. A ferrocenyl (Fc) group was attached to the hemilabile ligand to provide the redox center which can further assist and control the ring opening via an increment in the electrostatic repulsion of the two metallic centers. Thus, the weak-link is displaced by exogenous ligands only after the Fc group was oxidized.

Bond strengths for the hemilabile bonds were calculated at the ω-B97XD/lanl2dz level of theory upon optimized structures. Relative energies were calculated through the thermochemistry analysis (freq=noraman) made by Gaussian09 and the bond strengths were calculated with the NBODel procedure included in NBO3.1. In the open configurations we found that upon oxidation of Fc the exogenous ligand bond to Pt(II) strengthens by a few kcal/mol (2 – 10), however the Fe(III)-P distance increases and that can be observed via ³¹P NMR spectroscopy.
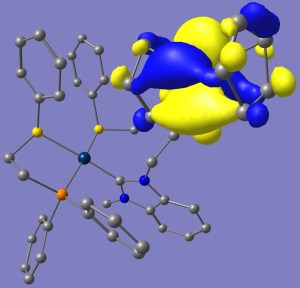
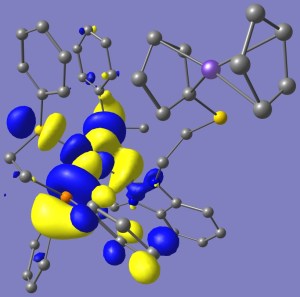
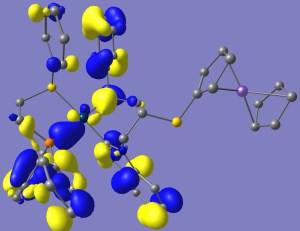
For the non-oxidized complexes, the HOMO’s are largely composed of the ferrocene highest energy orbitals, which is susceptible of being oxidized, whereas the LUMO’s are located throughout the organometallic fragment. When Ferrocene is oxidized to Ferrocenium, the situation is reversed and now HOMO’s are found spread over the organometallic fragment and the LUMO’s over ferrocenium; all of which is coherent with the idea of Fc now being able to be reduced. Plots for the HOMO LUMO orbitals for compound (6) in the Reduced (Fe2) and Oxidized (Fe3) states are shown (alpha and beta density are shown separately in the latter case).
Thanks to Prof. Chad Mirkin, Dr. Andrea d’Aquino, and Edmund Cheng for letting me be a part of this project.
[1] D’Aquino, A. I., Cheng, H. F., Barroso-Flores, J., Kean, Z. S., Mendez-Arroyo, J., McGuirk, C. M., & Mirkin, C. A. (2018). An Allosterically Regulated, Four-State Macrocycle. Inorganic Chemistry, 57(7), 3568–3578.
[2] Mendez-Arroyo, J., Barroso-Flores, J., Lifschitz, A. M., Sarjeant, A. a., Stern, C. L., & Mirkin, C. a. (2014). A multi-state, allosterically-regulated molecular receptor with switchable selectivity. Journal of the American Chemical Society, 136(29), 10340–10348.