I don’t know why I haven’t written about the Local Bond Order (LBO) before! And a few days ago when I thought about it my immediate reaction was to shy away from it since it would constitute a blatant self-promotion attempt; but hell! this is my blog! A place I’ve created for my blatant self-promotion! So without further ado, I hereby present to you one of my own original contributions to Theoretical Chemistry.
During the course of my graduate years I grew interested in weakly bonded inorganic systems, namely those with secondary interactions in bidentate ligands such as xanthates, dithiocarboxylates, dithiocarbamates and so on. Description of the resulting geometries around the central metallic atom involved the invocation of secondary interactions defined purely by geometrical parameters (Alcock, 1972) in which these were defined as present if the interatomic distance was longer than the sum of their covalent radii and yet smaller than the sum of their van der Waals radii. This definition is subject to a lot of constrictions such as the accuracy of the measurement, which in turn is related to the quality of the monocrystal used in the X-ray difraction experiment; the used definition of covalent radii (Pauling, Bondi, etc.); and most importantly, it doesn’t shed light on the roles of crystal packing, intermolecular contacts, and the energetics of the interaction.
This is why in 2004 we developed a simple yet useful definition of bond order which could account for a single molecule in vacuo the strength and relevance of the secondary interaction, relative to the well defined covalent bonds.
Barroso-Flores, J. et al. Journal of Organometallic Chemistry 689 (2004) 2096–2102 http://dx.doi.org/10.1016/j.jorganchem.2004.03.035,
Let a Molecular Orbital be defined as a wavefunction ψi which in turn may be constructed by a linear combination of Atomic Orbitals (or atom centered basis set functions) φj
We define ζLBO in the following way, where we explicitly take into account a doubly occupied orbital (hence the multiplication by 2) and therefore we are assuming a closed shell configuration in the Restricted formalism.
The summation is carried over all the orbitals which belong to atom A1 and those of atom A2.
Simplifying we yield,
where Sjk is the overlap integral for the φj and φk functions.
By summing over all i MOs we have accomplished with this definition to project all the MO’s onto the space of those functions centered on atoms A1 and A2. This definition is purely quantum mechanical in nature and is independent from any geometric requirement of such interacting atoms (i.e. interatomic distance) thus can be used as a complement to the internuclear distance argument to assess the interaction between them. This definition also results very simple and easy to calculate for all you need are the coefficients to the LCAO expansion and the respective overlap integrals.
Unfortunately, the Local Bond Order hasn’t found much echo, partly due to the fact that it is hidden in a missapropriate journal. I hope someone finds it interesting and useful; if so, don’t forget to cite it appropriately 😉


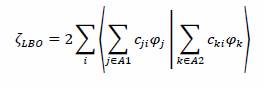
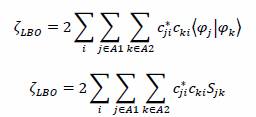
Hello colleagues, its impressive piece of writing concerning teachingand fully explained, keep iit uup all
the time.