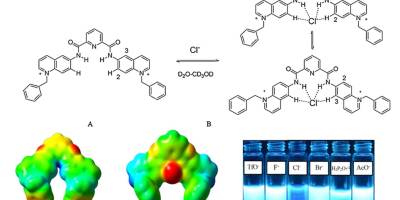
Chemosensors, Computational Chemistry, Paper, Sensors
9 Posts

Chemosensors, Computational Chemistry, Paper, Sensors

CCIQS, IQUNAM, Photosysnthesis, Talks

Articles, Fluorescence, Fluorescence Quenching, Paper

#RealTimeChem, #RealTimeChemCarnival, Computational Chemistry, DNA, RealTimeChem, Research, Twitter

#RealTimeChem, #RealTimeChemCarnival, Chemistry, Drug Delivery, Food, Science, Talks, Teaching, Twitter

#RealTimeChem, #RealTimeChemCarnival, Chemistry, RealTimeChem, Teaching, Twitter
#RealTimeChem, #RealTimeChemCarnival, Blogging, Chemistry, Computational Chemistry, Twitter

#RealTimeChem, Blogging, Chemistry, COMECyT, Computational Chemistry, Internet, RealTimeChem, Teaching, Theoretical Chemistry, Twitter

#RealTimeChem, Blogging, Chemistry, Computational Chemistry, Internet, Research, Twitter