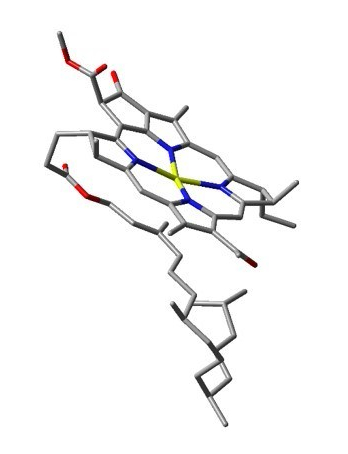
Photosynthesis, the basis of life on Earth, is based on the capacity a living organism has of capturing solar energy and transform it into chemical energy through the synthesis of macromolecules like carbohydrates. Despite the fact that most of the molecular processes present in most photosynthetic organisms (plants, algae and even some bacteria) are well described, the mechanism of energy transference from the light harvesting molecules to the reaction centers are not entirely known. Therefore, in our lab we have set ourselves to study the possibility of some excitonic transference mechanisms between pigments (chlorophyll and its corresponding derivatives). It is widely known that the photophysical properties of chlorophylls and their derivatives stem from the electronic structure of the porphyrin and it is modulated by the presence of Mg but its not this ion the one that undergoes the main electronic transitions; also, we know that Mg almost never lies in the same plane as the porphyrin macrocycle because it bears a fifth coordination whether to another pigment or to a protein that keeps it in place (Figure 1).

During our calculations of the electronic structure of the pigments (Bacteriochlorophyll-a, BChl-a) present in the Fenna-Matthews-Olson complex of sulfur dependent bacteria we found that the Mg²⁺ ion at the center of one of these pigments could in fact create an intermolecular interaction with the C=C double bond in the phytol fragment which lied beneath the porphyrin ring.
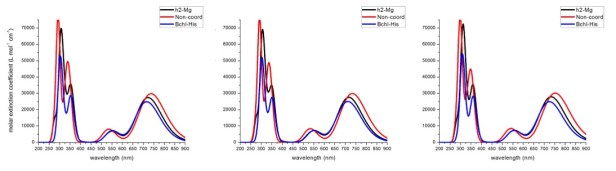
This would be the first time that a dihapto coordination is suggested to occur in any chlorophyll and that on itself is interesting enough but we took it further and calculated the photophysical implications of having this fifth intramolecular dihapto coordination as opposed to a protein or none for that matter. Figure 3 shows that the calculated UV-Vis spectra (calculated with Time Dependent DFT at the CAM-B3LYP functional and the cc-pVDZ, 6-31G(d,p) and 6-31+G(d,p) basis sets). A red shift is observed for the planar configuration, respect to the five coordinated species (regardless of whether it is to histidine or to the C=C double bond in the phytyl moiety).
Before calculating the UV-Vis spectra, we had to unambiguously define the presence of this observed interaction. To that end we calculated to a first approximation the C-Mg Wiberg bond indexes at the CAM-B3LYP/cc-pVDZ level of theory. Both values were C(1)-Mg 0.022 and C(2)-Mg 0.032, which are indicative of weak interactions; but to take it even further we performed a non-covalent interactions analysis (NCI) under the Atoms in Molecules formalism, calculated at the M062X density which yielded the presence of the expected critical points for the η²Mg-(C=C) interaction. As a control calculation we performed the same calculation for Magnoscene just to unambiguously assign these kind of interactions (Fig 4, bottom).

This research is now available at the International Journal of Quantum Chemistry. A big shoutout and kudos to Gustavo “Gus” Mondragón for his work in this project during his masters; many more things come to him and our group in this and other research ventures.

i think that you give the scientific community a good work
Thank you, Hadji!