Well, I only contributed with the theoretical section by doing electronic structure calculations, so it isn’t really a paper we can ascribe to this particular lab, however it is really nice to see my name in JACS along such a prominent researcher as Prof. Chad Mirkin from Northwestern University, in a work closely related to my area of research interest as macrocyclic recognition agents.
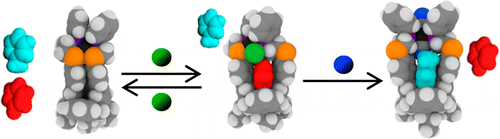
In this manuscript, a calix[4]arene is allosterically opened and closed reversibly by coordinating different kinds of ligands to a platinum center linked to the macrocycle. (This approach has been referred to as the weak link approach.) I recently visited Northwestern and had a great time with José Mendez-Arroyo, the first author, who showed me around and opened the possibility for further work between our research groups.
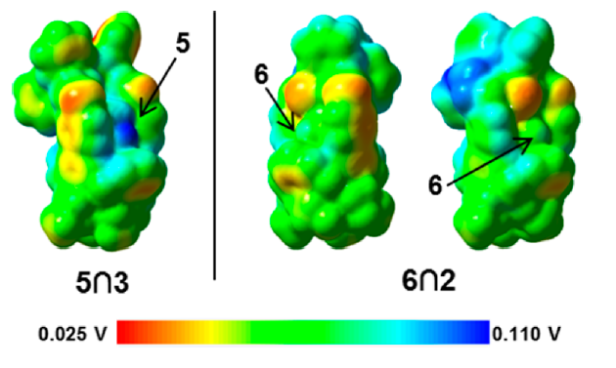
Closed, semi-open and fully open conformations; selectivity is modulated through cavity size. (Ligands: Green = Chloride; Blue = Cyanide)
Here at UNAM we calculated the interaction energies for the two guests that were successfully inserted into the cavity: N-methyl-pyridinium (Eint = 57.4 kcal/mol) and Pyridine-N-oxide (Eint = +200.0 kcal/mol). Below you can see the electrostatic potential mapped onto the electron density isosurface for one of the adducts. Relative orientation of the hosts within the cavity follows the expected (anti-) alignment of mutual dipole moments. At this level of theory, we could easily be inclined to assert that the most stable interaction is indeed the one from the semi-open compound and that this in turn is due to the fact that host and guest are packed closer together but there is also an orbital issue: Pyridine Oxide is a better electron acceptor than N-Me-pyridinium and when we take a closer look to the (Natural Bonding) orbitals interacting it becomes evident that a closer location does not necessarily yields a stronger interaction when the electron accepting power of the ligand is weaker (which is, in my opinion, both logic and at the same time a bit counterintuitive, yet fascinating, nonetheless).
All calculations were performed at the B97D/LANL2DZ level of theory with the use of Gaussian09 and NBO3.1 as provided within the former. Computing time at UNAM’s supercomputer known as ‘Miztli‘ is fully acknowledged.
The full citation follows:
A Multi-State, Allosterically-Regulated Molecular Receptor With Switchable Selectivity
Jose Mendez-Arroyo †, Joaquín Barroso-Flores §,Alejo M. Lifschitz †, Amy A. Sarjeant †, Charlotte L. Stern †, and Chad A. Mirkin *†
Thanks to José Mendez-Arroyo for contacting me and giving me the opportunity to collaborate with his research; I’m sure this is the first of many joint projects that will mutually benefit our groups.

I appreciate that I share some of your projects that carry out so give me a better idea of the computer packages that used for the simulations, I’m entering myself a little molecular structures to complement fieldwork that I conducted with physical plasma thank you very much greetings.
El Jueves, 10 de julio, 2014 9:20:24, Dr. Joaquin Barroso’s Blog escribió:
WordPress.com joaquinbarroso posted: “Well, I only contributed with the theoretical section by doing electronic structure calculations, so it isn’t really a paper we can ascribe to this particular lab, however it is really nice to see my name in JACS along such a prominent researcher as Prof.”
Thanks a lot dr BARROSO for what you are doing to make theoritical chemistry chine. I need you helpe on this problem I face since almost one year in my works without a hope of solution. It concerns an IRC calculation. Here is the link I use with G09 followed by the the message I read in the output file
Link
%nprocshared=8
%mem=120MW
# irc=(maxpoints=50,calcfc,stepsize=5) b3lyp/lanl2dz pop=nbo
geom=connectivity
Output file mention
Cartesian Forces: Max 0.001970780 RMS 0.000610537
IRC-IRC-IRC-IRC-IRC-IRC-IRC-IRC-IRC-IRC-IRC-IRC-IRC-IRC-IRC-IRC-IRC-IRC
Error in corrector energy = -0.0000715184
Magnitude of corrector gradient = 0.0045854547
Magnitude of analytic gradient = 0.0052874022
Magnitude of difference = 0.0057829139
Angle between gradients (degrees)= 71.3071
Pt 35 Step number 20 out of a maximum of 20
Modified Bulirsch-Stoer Extrapolation Cycles:
EPS = 0.000010000000000
Maximum DWI energy std dev = 0.000006827 at pt -1
Maximum DWI gradient std dev = 0.296154925 at pt 64
CORRECTOR INTEGRATION CONVERGENCE:
Recorrection delta-x convergence threshold: 0.010000
Delta-x Convergence NOT Met
Maximum number of corrector steps exceded.
Error termination via Lnk1e in /usr/local/gaussian/g09/l123.exe at Sun Jul 13 10:31:58 2014.
I would like to ask any person on the blog who can help to answer me.
Hi dr
I’m going to run code NBO 5 in g09 or g03 but I can not? or convert NBO3.1 to NBO 5? I would buy this code.
hi sir while i am optimizing 24 atoms file on Gaussian 03 but i am getting the following error
dumping /fiocom/, unit = 3 NFiles = 1 SizExt = 524288 WInBlk = 512
defal = T LstWrd = 67072 FType=2 FMxFil=10000
Number 0
Base 20480
End 67072
End1 67072
Wr Pntr 20480
Rd Pntr 20480
Length 46592
Error termination in NtrErr:
NtrErr called from FIOCnC.
by using the following path
%mem=1gb
# opt=(calcall) mp2/6-311++g(d,p) scf=(qc,maxcycle=5000)
what wrong i dont know how to over come plz tell me sir
Sir, I ran a Gaussian work in Gaussian with the input
# irc=(calcfc maxpoints=20) hf/6-31g(d) scrf test
But ended with this out put Angle between gradients (degrees)= 64.9741
Pt 15 Step number 20 out of a maximum of 20
CORRECTOR INTEGRATION CONVERGENCE:
Recorrection delta-x convergence threshold: 0.010000
Delta-x Convergence NOT Met
Maximum number of corrector steps exceded.
Error termination via Lnk1e in C:\G09W\l123.exe at Sat Jan 09 23:07:16 2016.
How can I got the correct route section?
Partha