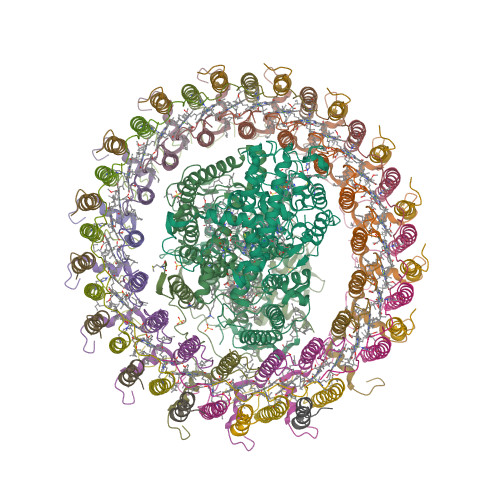
Photosynthetic organisms are so widespread around the globe they have adapted to various solar lighting conditions to thrive. The bacteria Blastochloris viridis absorbs light in the near infrared region of the electromagnetic spectrum, in fact, it holds the record for the longest wavelength (~1015 nm) absorbing organism whose Light Harvesting complex 1 (LHC1) has been elucidated. Despite their adaptation to a wide number of light conditions, photosynthetic organism can only make use of so many pigments or chromophores; the LHC1 (Figure 1) in B. viridis in fact is made up of Bacteriochlorophyll-b (BChl-b) molecules, one of the most abundant photosynthetic pigments on Earth, whose main absorption in solution (MeOH) is observed at 795 nm.
So, how can B. viridis use BChl-b molecules to absorb near IR radiation and how does it achieve this remarkable red-shifting effect? The LHC1 structure was solved in 2018 by Qian et al. through Cryo-EM at a 2.9 Å resolution; it is comprised of 17 protein subunits surrounding the so called photosynthetic pigments special pair. Each of these subunits is made up of three α-helix structures surrounding two BChl-b and one dihydroneurosporene (DHN) molecule for a total of 34 of these photosynthetic pigments inside the LHC and 17 DHN molecules interacting between the protein structures and the
main BChl-b pigments.
It was Dr. Jacinto Sandoval and Gustavo “Gus” Mondragón who brought this facts to our attention during their survey of potential candidates for calculating exotic exciton transfer mechanisms in photosynthetic organisms, part of Gustavo’s PhD thesis. To them, it was clear from the start that some sort of cooperative effect between pigments was operating and possibly leading to the red-shifted absorption, therefore a careful dissection of all possible pigments combinations was carried out and their UV-Vis spectra were calculated at the CAMB3LYP/cc-pVDZ on PBE0/6-31G(d) optimized geometries, leading to the systems shown below in figure 2.

System B7 reproduced the red-shifted absorption at 1026 nm, but since the original structure was fitted from the Cryo-EM with a 2.9 Å resolution, “Gus” suggested reaching out to the group of Prof. Andrés Gerardo Cisneros and Dr. Jorge Nochebuena at UT Dallas for carrying out QM/MM calculations; this optimization included the proteins surrounding the pigments in the MM layer and the interacting residues (Hys coordinated to Mg2+ ions in BChl-b) along the chromophores were incorporated into the QM layer, however the thus obtained minima for the B7 system lost the main absorption in the near-IR region, therefore, Dr. Nochebuena suggested running an MD simulation (45 ns) and took a random sampling of ten structures (Figure 3).
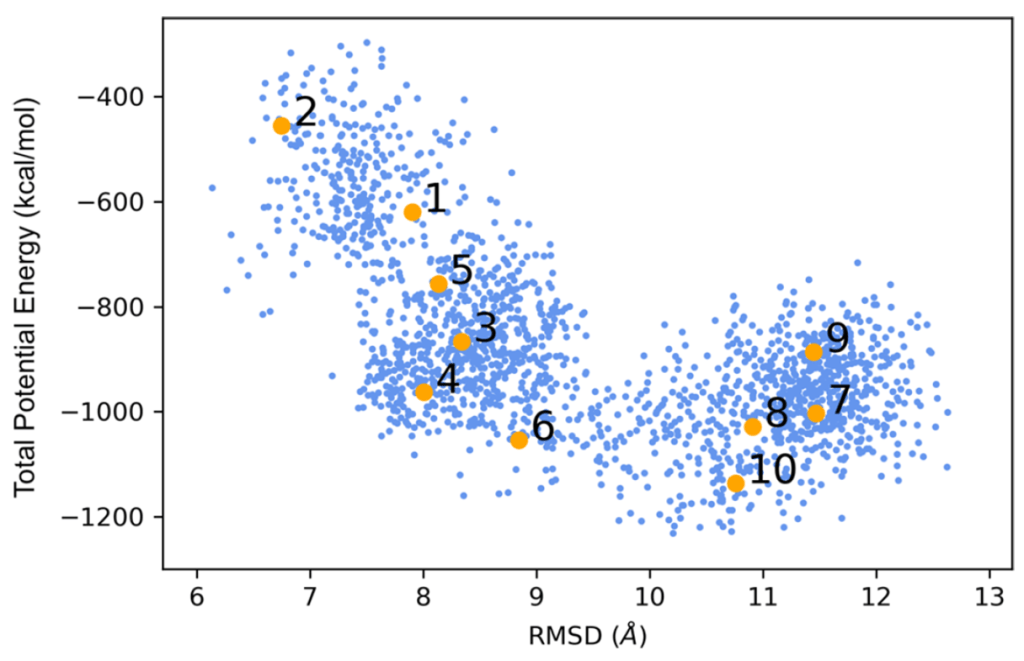
clustering analysis.
All structures in the sampling reproduced the red-shifted absorption (~1000 nm) successfully proving that cooperative and dynamic effects allow B. viridis to perform photosynthesis with low energy radiation (Figure 4). Therefore, close intermolecular interactions along with thermal/dynamical fluctuations allow for a regular pigment such as BChl-b to form near-IR absorbing photosystems for organisms to thrive in low conditions of solar light.
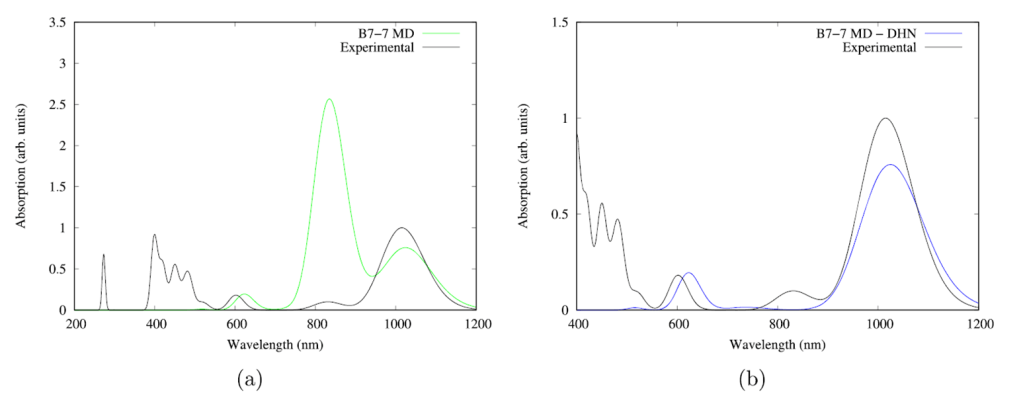
molecule fragment. (b) Calculated spectrum (blue) excluding the DHN molecule fragment.
If you want to read further details, this work is now published in the Journal of Chemical Theory and Computation of the American Chemical Society. I’ll talk about this and other ventures in photosynthesis next week at the WATOC conference in Vancouver, swing by to talk CompChem!

Thanks, very interesting !
Ionel Haiduc