If you work in the field of photovoltaics or polyacene photochemistry, then you are probably aware of the Singlet Fission (SF) phenomenon. SF can be broadly described as the process where an excited singlet state decays to a couple of degenerate coupled triplet states (via a multiexcitonic state) with roughly half the energy of the original singlet state, which in principle could be centered in two neighboring molecules; this generates two holes with a single photon, i.e. twice the current albeit at half the voltage (Fig 1).
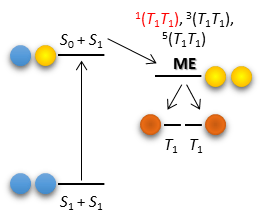
It could also be viewed as the inverse process to triplet-triplet annihilation. An important requirement for SF is that the two triplets to which the singlet decays must be coupled in a 1(TT) state, otherwise the process is spin-forbidden. Unfortunately (from a computational perspective) this also means that the 3(TT) and 5(TT) states are present and should be taken into account, and when it comes to chlorophyll derivatives the task quickly scales.
SF has been observed in polyacenes but so far the only photosynthetic pigments that have proven to exhibit SF are some carotene derivatives; so what about chlorophyll derivatives? For a -very- long time now, we have explored the possibility of finding a naturally-occurring, chlorophyll-based, photosynthetic system in which SF could be possible.
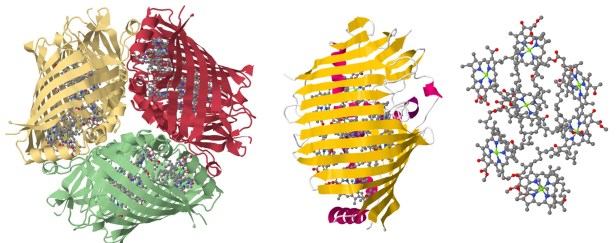
But first things first; The methodology: It was soon enough clear, from María Eugenia Sandoval’s MSc thesis, that TD-DFT wasn’t going to be enough to capture the whole description of the coupled states which give rise to SF. It was then that we started our collaboration with SF expert, Prof. David Casanova from the Basque Country University at Donostia, who suggested the use of Restricted Active Space – Spin Flip in order to account properly for the spin change during decay of the singlet excited state. A set of optimized bacteriochlorophyll-a molecules (BChl-a) were oriented ad-hoc so their Qy transition dipole moments were either parallel or perpendicular; the rate to which SF could be in principle present yielded that both molecules should be in a parallel Qy dipole moments configuration. When translated to a naturally-occurring system we sought in two systems: The Fenna-Matthews-Olson complex (FMO) containing 7 BChl-a molecules and a chlorosome from a mutant photosynthetic bacteria made up of 600 Bchl-d molecules (Fig 2). The FMO complex is a trimeric pigment-protein complex which lies between the antennae complex and the reaction center in green sulfur dependent photosynthetic bacteria such as P. aestuarii or C. tepidium, serving thus as a molecular wire in which is known that the excitonic transfer occurs with quantum coherence, i.e. virtually no energy loss which led us to believe SF could be an operating mechanism. So far it seems it is not present. However, for a crystallographic BChl-d dimer present in the chlorosome it could actually occur even when in competition with fluorescence.

I will keep on blogging more -numerical and computational- details about these results and hopefully about its publication but for now I will wrap this post by giving credit where credit is due: This whole project has been tackled by our former lab member María Eugenia “Maru” Sandoval and Gustavo Mondragón. Finally, after much struggle, we are presenting our results at WATOC 2017 next week on Monday 28th at poster session 01 (PO1-296), so please stop by to say hi and comment on our work so we can improve it and bring it home!

Dear Joaquin,
I think that in your “Jablonski’s Diagram for SF”, the lowest levels should be “S0 + S0”.
Best regards,
Clovis.
Thanks for pointing it out, Clovis. You’re absolutely right, I’ve fixed this figure in other instances but not here apparently. I’ll get to it eventually.
Best regards
Hi professor Barroso. Do you have any post, or can you recommend me any resource on how to calculate the Qy transition dipole moment? I have some knowledge in Q-chem and Nwchem, but materials in other software in general would be helpful. Thanks for your help.